|
Continuous spectra are produced by all incandescent solids and liquids and by gases under high pressure. The spectrum formed from white light contains all colors, or frequencies, and is known as a continuous spectrum. In addition to visible light, other types of electromagnetic radiation may be spread into a spectrum according to frequency or wavelength. Each different wavelength or frequency of visible light corresponds to a different color, so that the spectrum appears as a band of colors ranging from violet at the short-wavelength (high-frequency) end of the spectrum through indigo, blue, green, yellow, and orange, to red at the long-wavelength (low-frequency) end of the spectrum. Continuous and Line Spectraĭispersion, the separation of visible light into a spectrum, may be accomplished by means of a prism or a diffraction grating. Physicists often find it useful to separate a beam of particles into a spectrum according to their energy. Beams of charged particles can be separated into a spectrum according to mass in a mass spectrometer (see mass spectrograph). 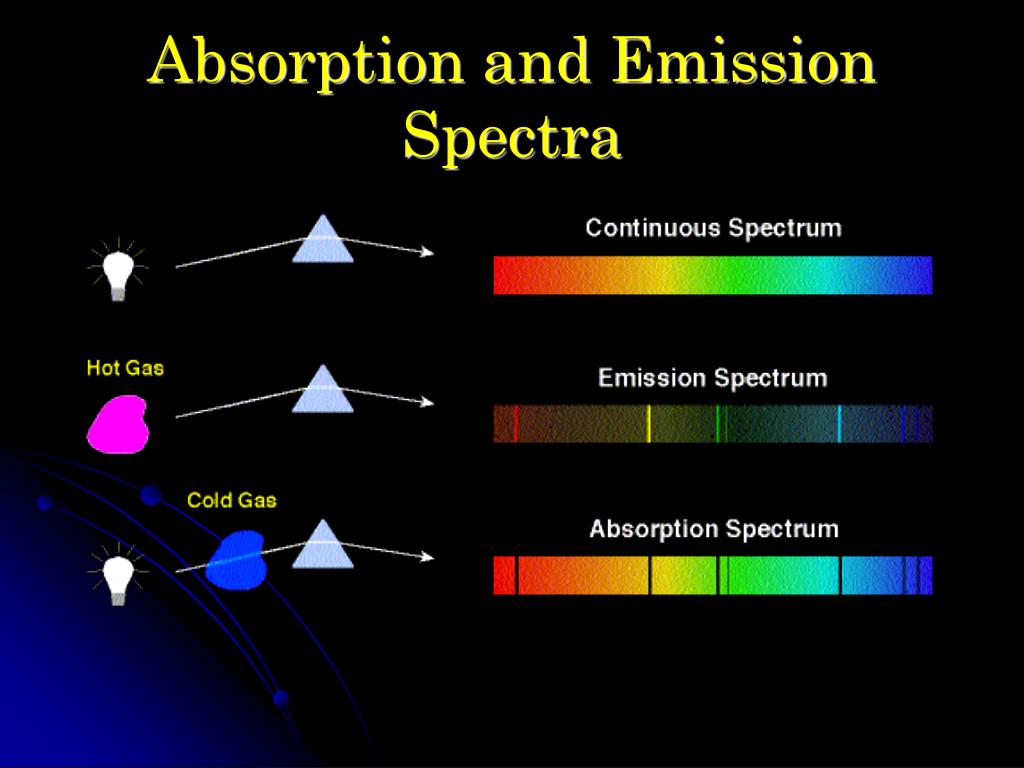
Since a colour can be described as a specific frequency of light, this is why the black breaks can be used to identify element(s) which the light is passing through.Spectrum, arrangement or display of light or other form of radiation separated according to wavelength, frequency, energy, or some other property. Where E is the energy of a photon, h is Planck's constant (a constant is a set number that doesn't change) and f is the frequency of the photon. The energy of a photon is proportional to its frequency: Note that the energy gained by the excited electron is equal to the energy of the incident photon (a particle of light), so only photons with fitting energies will cause an excitation. The missing colours give us information about the energy of the photons that cause excitation. We can then reach a conclusion about what the distant planet's atmosphere may be made of. This is important because it allows us to compare the absorption lines of say, the atmosphere of a far away planet, to the absorption lines of elements we know to exist in a lab. For any atom of a particular element, the energy needed to excite an electron from one specific energy level to another will be the same. The process of an electron going to a higher energy level is called excitation. From experiments, electrons only ever absorb certain amounts of energy, suggesting an electron's energy must fit onto set, quantised, discrete energy levels. On an atomic scale, this happens because of the electrons in the atoms of the sample - an electron can absorb light to gain energy. These breaks are called absorption lines, and every element has its characteristic pattern of absorption lines. An image is created of the spectrum of light with black breaks where the light has been absorbed. These colours of light are being absorbed by the sample. 
When a full spectrum of light (light with all the colours, like light from the sun) passes through the sample (which is often a gas) some specific colours do not show up on the other side. 
Absorption spectroscopy is a technique used to find out what makes up a sample of a substance – in other words, a chemical analysis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
